Description
Dimethyl sulfoxide (DMSO) is a key dipolar aprotic solvent. It is less toxic than other members of this class: dimethylformamide, dimethylacetamide, N-methyl-2-pyrrolidone, HMPA. Dimethyl sulfoxide is the chemical compound (CH3)2SO. This colorless liquid is an important dipolar aprotic solvent. It is readily miscible in a wide range of organic solvents as well as water. It has a distinctive property of penetrating the skin very readily, allowing the handler to taste it. Some describe it as an “oyster-like” taste, others claim it tastes like garlic. DMSO is also employed as a rinsing agent in the electronics industry and, in its deuterated form (DMSO-d6), is a useful solvent in NMR due to its ability to dissolve a wide range of chemical compounds and its minimal interference with the sample signals. In cryobiology DMSO has been used as a cryoprotectant and is still an important constituent of cryoprotectant vitrification mixtures used to preserve organs, tissues, and cell suspensions. It is particularly important in the freezing and long-term storage of embryonic stem cells and hematopoietic stem cell, which are often frozen in a mixture of 10% DMSO and 90% fetal calf serum. As part of an autologous bone marrow transplant the DMSO is re-infused along with the patient’s own hematopoietic stem cell. Dimethyl sulfoxide is a by-product of wood pulping. One of the leading suppliers of DMSO is the Gaylord company in the USA. DMSO is frequently used as solvent in a number of chemical reactions. In particular it is an excellent reaction solvent for SN2 alkylations: it is possible to alkylate indoles with very high yields using potassium hydroxide as the base and a similar reaction also occurs with phenols. DMSO can be reacted with methyl iodide to form a sulfoxonium ion which can be reacted with sodium hydride to form a sulfur ylide. The methyl groups of DMSO are somewhat acidic in character (pKa=35) due to the stabilization of the resultant anions by the sulfoxide group.
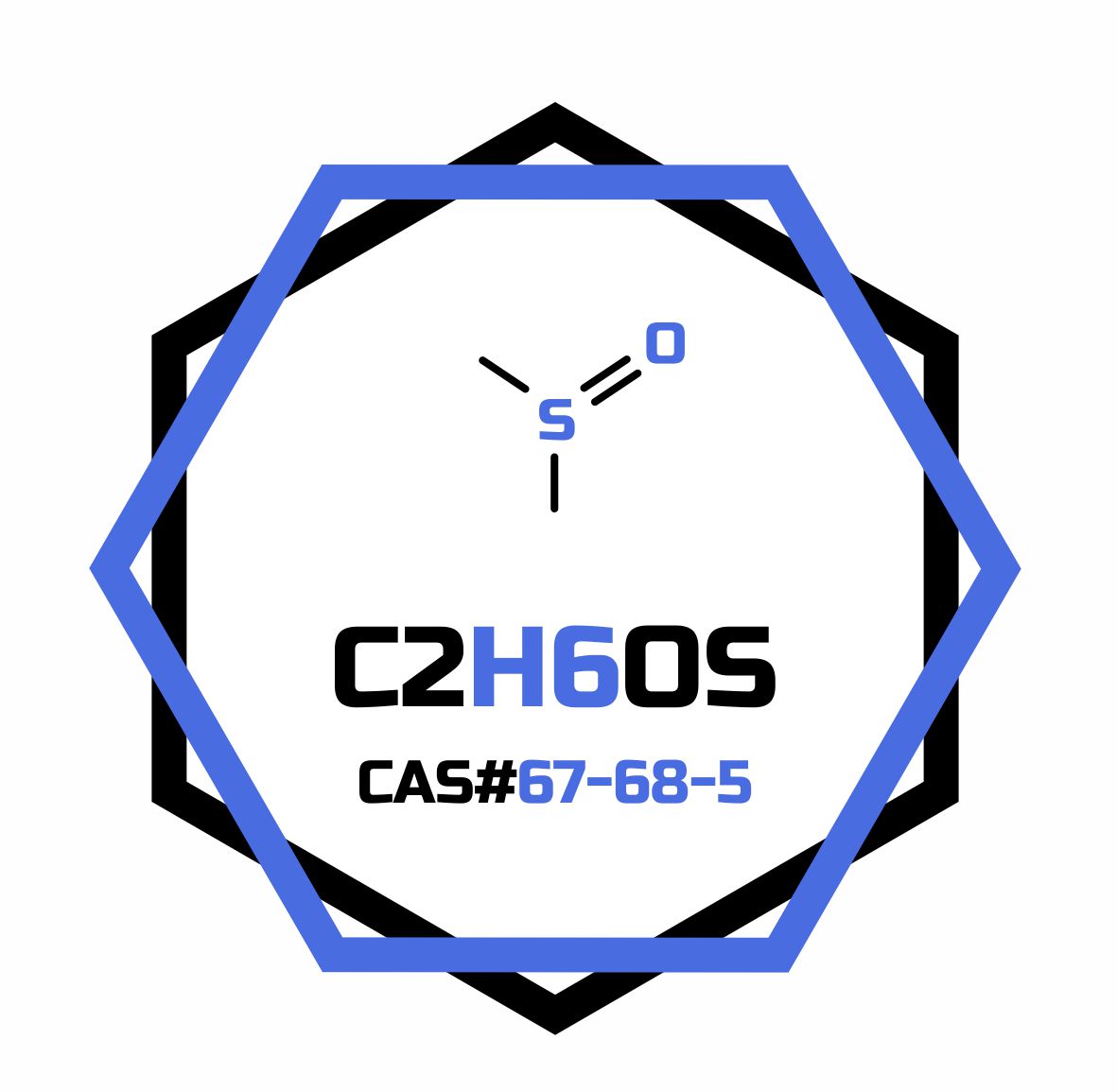
- Formula
- C2H6OS
- Molar mass
- 78.13 g/mol
- CAS Number
- 67-68-5
Reviews
There are no reviews yet.