Description
Cyclohexylamine is a family of alicyclic hydrocarbons containing an amine group with the general formula R-C6H10NH2. Cyclohexylamine is a food contaminant arising from its use as a boiler water additive Cyclohexylamine, also called hexahydroaniline, 1-aminocyclohexane, or aminohexahydrobenzene, is an organic chemical, an amine derived from cyclohexane. It is a clear to yellowish liquid with fishy odor, with melting point of -17. 7 C and boiling point 134. 5 C, miscible with water. Like other amines it is of mildly alkaline nature, compared to strong bases, such as NaOH, but it is a stronger base than its aromatic sister compound aniline (which differs only in that its ring is aromatic). It is flammable, with flash point at 28. 6 C. Explosive mixtures with air can be formed above 26 C. It is toxic by both ingestion and inhalation, the inhalation itself may be fatal. It readily absorbs through skin which it irritates. It is corrosive. Cyclohexylamine is listed as an extremely hazardous substance as defined by Section 302 of the U. S. Emergency Planning and Community Right-to-Know Act. Cyclohexylamine belongs to the family of Cycloalkanes. These are alkanes containing one or more rings of carbon atoms.
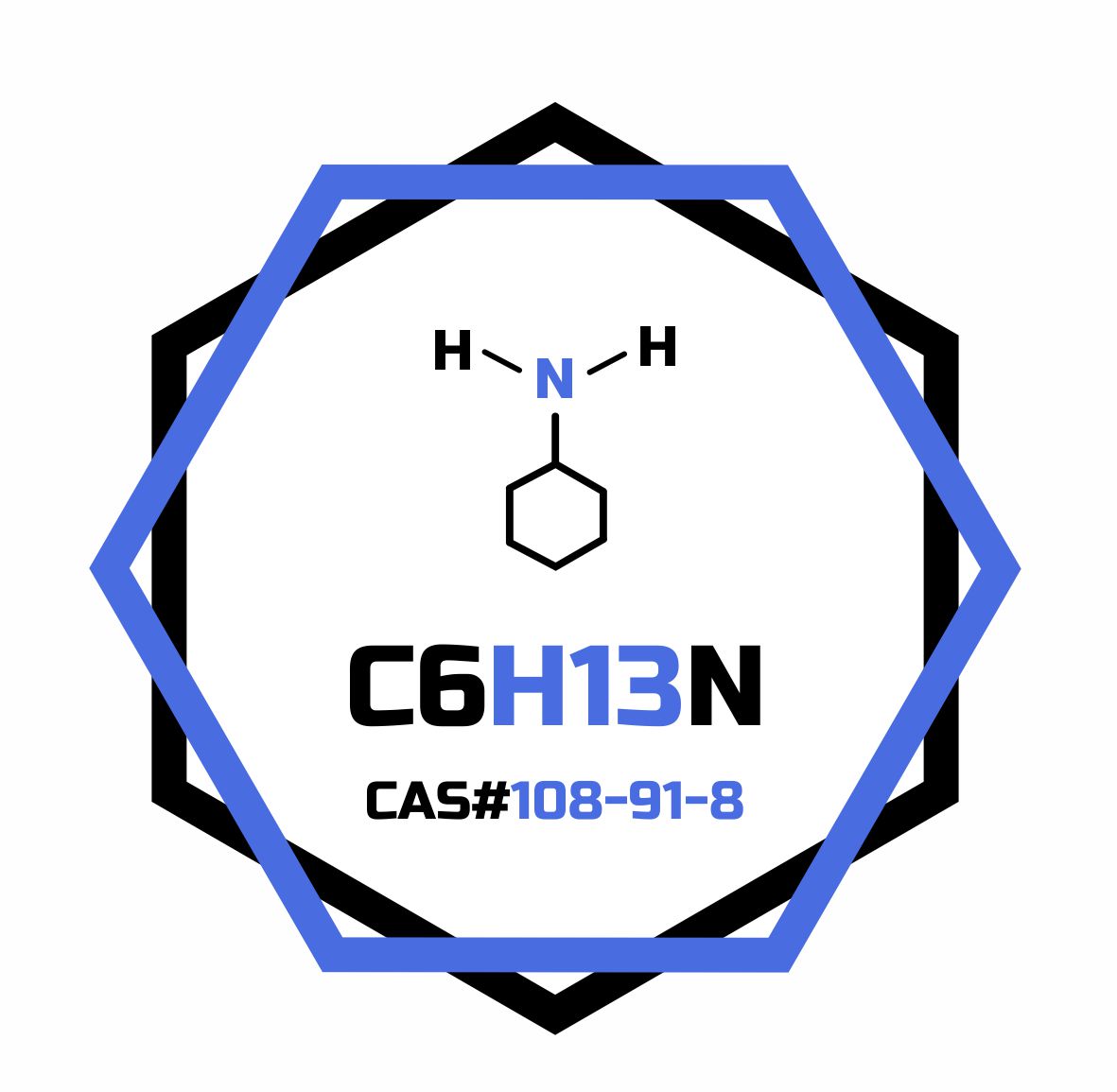
- Formula
- C6H13N
- Molar mass
- 99.177 g/mol
- CAS Number
- 108-91-8
- Density
- 0.8647 at 25°C (77°F)
- Purity/Grade
- 99%
- Apearance
- Liquid
- Melting point/freezing point
- -17.7°C
- Initial boiling point and boiling range
- 134.5°C
- Solubility in water
- Miscible
- Solubility in ethanol
- Very soluble
Note: The chemical specifications for some items on this product page may differ from the specifications listed above. For more information, consult with our manager. A detailed specification for any chemical product is available on request.
Reviews
There are no reviews yet.